Real-Time Cell Migration Monitoring Through Porous Membranes Using Dual-Sided Impedance Spectroscopy
NIST researchers developed CoCM, the first microfluidic platform capable of tracking cell migration on both sides of a porous membrane simultaneously using label-free impedance spectroscopy. Here’s how it works, what they found, and what it means for organ-on-chip research.
Paper: Torres-Castro, Rane & Reyes · DOI: 10.1039/D5LC00898KJournal: Lab on a Chip (RSC)Institution: NIST Published: March 2, 2026 · Open Access (CC-BY)

The Problem: Why Existing Cell Migration Assays Fall Short
Cell migration through porous tissue interfaces plays a critical role in cancer metastasis, immune responses, wound healing, and embryonic development. Understanding these dynamics in real-time is essential for advancing microphysiological systems (MPS) and organ-on-chip models.
However, existing platforms for studying cell migration through porous membranes have fundamental limitations:
Commercial transwell-based analyzers (e.g., xCELLigence RTCA) place impedance electrodes on only one side of the membrane. Cells must be seeded in a gel layer and are detected only after they have migrated to the opposite surface. This means the departure dynamics, when and how cells begin crossing , are entirely missed.
TEER-based microfluidic devices measure bulk impedance across the entire membrane and both channels, which limits spatial resolution. They are designed for barrier integrity measurements, not for tracking individual migration events at the membrane interface.
Conventional impedance-integrated microfluidics place electrodes on solid surfaces (glass, silicon, plastic) at the floor or ceiling of channels , not on the membrane itself. This means they can only measure cells when they depart from or arrive at the electrodes, but not during the actual membrane-crossing process.
The Gap
No existing platform could independently measure cell impedance on both surfaces of a porous membrane, which is exactly where the migration process occurs. That’s the gap the NIST team set out to fill.
How CoCM Works: Dual-Sided Impedance Spectroscopy on a Porous Membrane
The key innovation in this paper is conceptually simple but technically challenging: pattern interdigitated gold electrodes on both sides of a thin porous membrane, then measure impedance independently on each surface.
Electrode design
The researchers patterned 50 µm-wide gold electrodes with 5 µm gaps on both sides of a 16 µm-thick track-etched PET membrane (8 µm pore diameter, 6×10⁴ pores/cm²). Using alignment marks, they ensured the top and bottom electrode patterns were coincident, allowing light to pass through for simultaneous optical imaging.
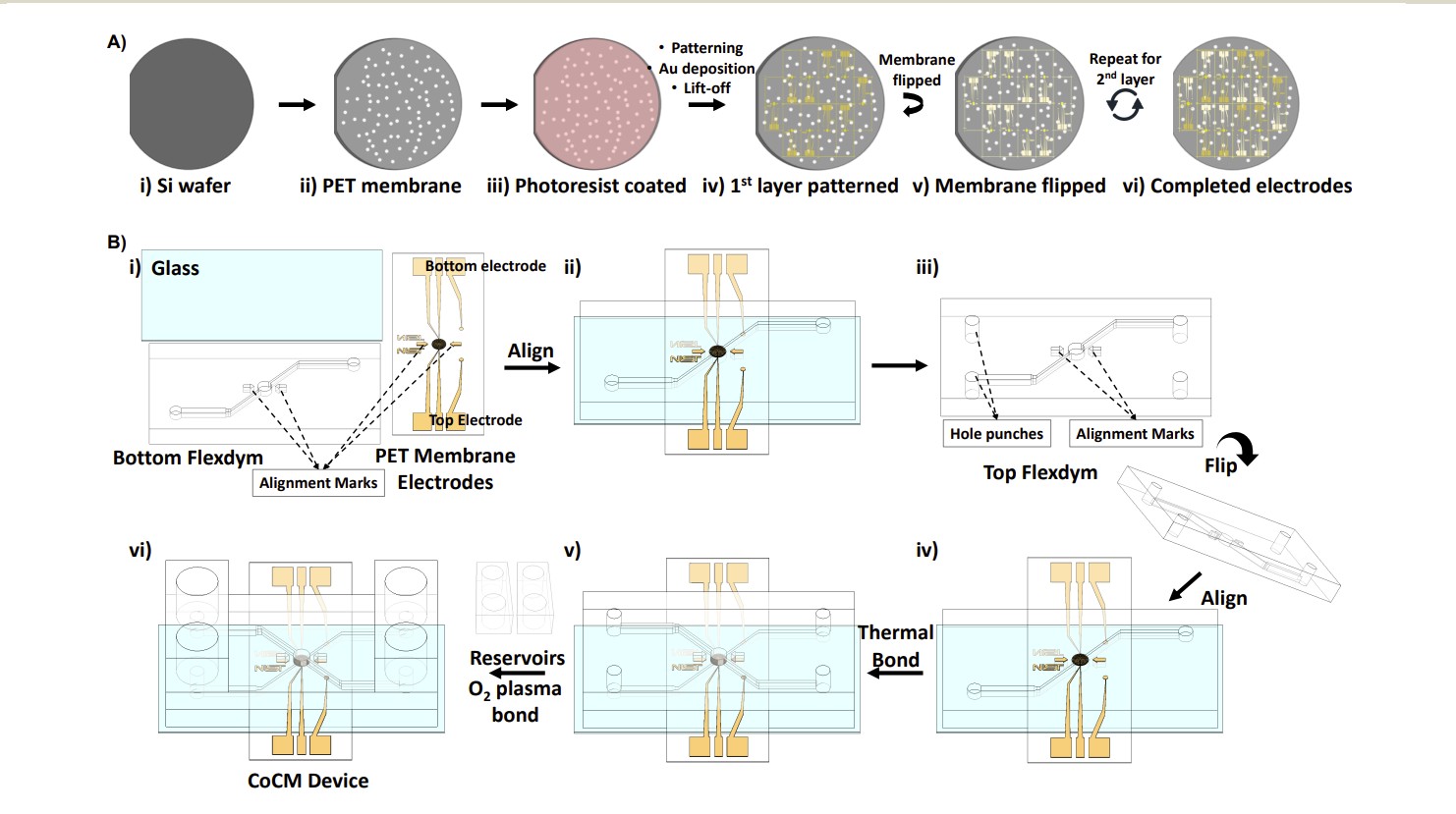
Figure 2. Photolithography workflow for patterning gold electrodes on both sides of a porous PET membrane. The membrane is placed on an hPDMS-coated silicon wafer, processed with standard lift-off photolithography, then flipped to repeat on the other side. From Torres-Castro et al., Lab Chip, 2026.
Measurement principle
Each side of the membrane has its own electrode pair connected to a separate potentiostat channel, operating at 30 kHz with measurements every 20 minutes. When cells adhere to or migrate across the electrodes, they modify the local impedance by altering the charge transfer resistance (Rct) and the constant phase element (CPE) capacitance at the electrode-electrolyte interface.
As cells leave the top surface, the top impedance decreases. As they arrive on the bottom surface, the bottom impedance increases. These inversely proportional, symmetrical trends constitute the CoCM signature.
The CoCM index: a normalization method
To enable direct comparison between two different potentiostats measuring two different electrode sets, the team developed a normalization method called the CoCM index. Each channel’s hourly impedance average is divided by its first-hour baseline, producing a dimensionless index starting at 1.0 that tracks relative changes over time. This accounts for variability in chip connections, electrode fabrication, and instrument offsets.

Equation 1. The CoCM index normalization, where i denotes the channel (top or bottom potentiostat), h is the hour, and n indexes individual measurements within that hour.
Device Fabrication: Materials and Methods
The CoCM device consists of two vertically aligned microfluidic chambers separated by the electrode-patterned PET membrane. Here is a summary of the fabrication process, including the Eden Microfluidics products used and complementary solutions that could streamline the workflow for other labs.
| Step | Process | What NIST Did | Eden Solution |
|---|---|---|---|
| 1 | Chip design | Custom design with two vertically aligned chambers, alignment marks, and inlet/outlet geometry. CFD simulation for shear stress optimization (average FSS: 0.034 Pa, max: 0.120 Pa at 5 µm above membrane). | Complementary FLUI’DEVICE — online design & flow simulation software |
| 2 | Mold fabrication | SU-8 negative photoresist on silicon wafer, creating ~100 µm high channels. Cleanroom photolithography process. | Complementary FLUI’MOLD — 3D-printed molds, no cleanroom required. EPOXYM for mold replication. |
| 3 | Hot embossing | Used in study SUBLYM hot embosser — Pattern transfer from SU-8 mold into FLEXDYM sheets at 170 °C for 360 s. | |
| 4 | Channel material | Used in study FLEXDYM™ thermoplastic copolymer — chosen for biocompatibility, low gas permeability, and thermal bonding at 80 °C. ISO 10993, USP Class VI certified. | |
| 5 | Assembly & bonding | Glass substrate → bottom FLEXDYM channel → PET membrane with electrodes → top FLEXDYM channel. Thermal bonding at 80 °C, 1 min. PDMS reservoirs O₂ plasma bonded to inlets/outlets. | Used SUBLYM also served as thermal bonding tool (2-in-1 embossing & bonding) |
Cell seeding and culture
786-O human renal carcinoma cells (ATCC) were stained with CellTracker Red and seeded at 2×10⁶ cells/mL in the top chamber using passive, gravity-driven flow. The membrane was pre-coated with fibronectin and polyallylamine hydrochloride (PAH) to promote adhesion. Media (RPMI-1640 + 10% FBS) was changed every 6–8 hours to compensate for evaporation and nutrient depletion.
(top chamber)
(bottom chamber)
at 70 h
CoCM trends
(cell-free)
CoCM index dynamics
Over 70 hours (n=3 replicates), the top chamber CoCM index progressively decreased as cells migrated away from the seeding surface, while the bottom chamber index increased as cells arrived and adhered. The most pronounced change occurred after approximately 40 hours, when larger cell clusters began crossing the membrane, producing clear symmetrical, inversely proportional trends
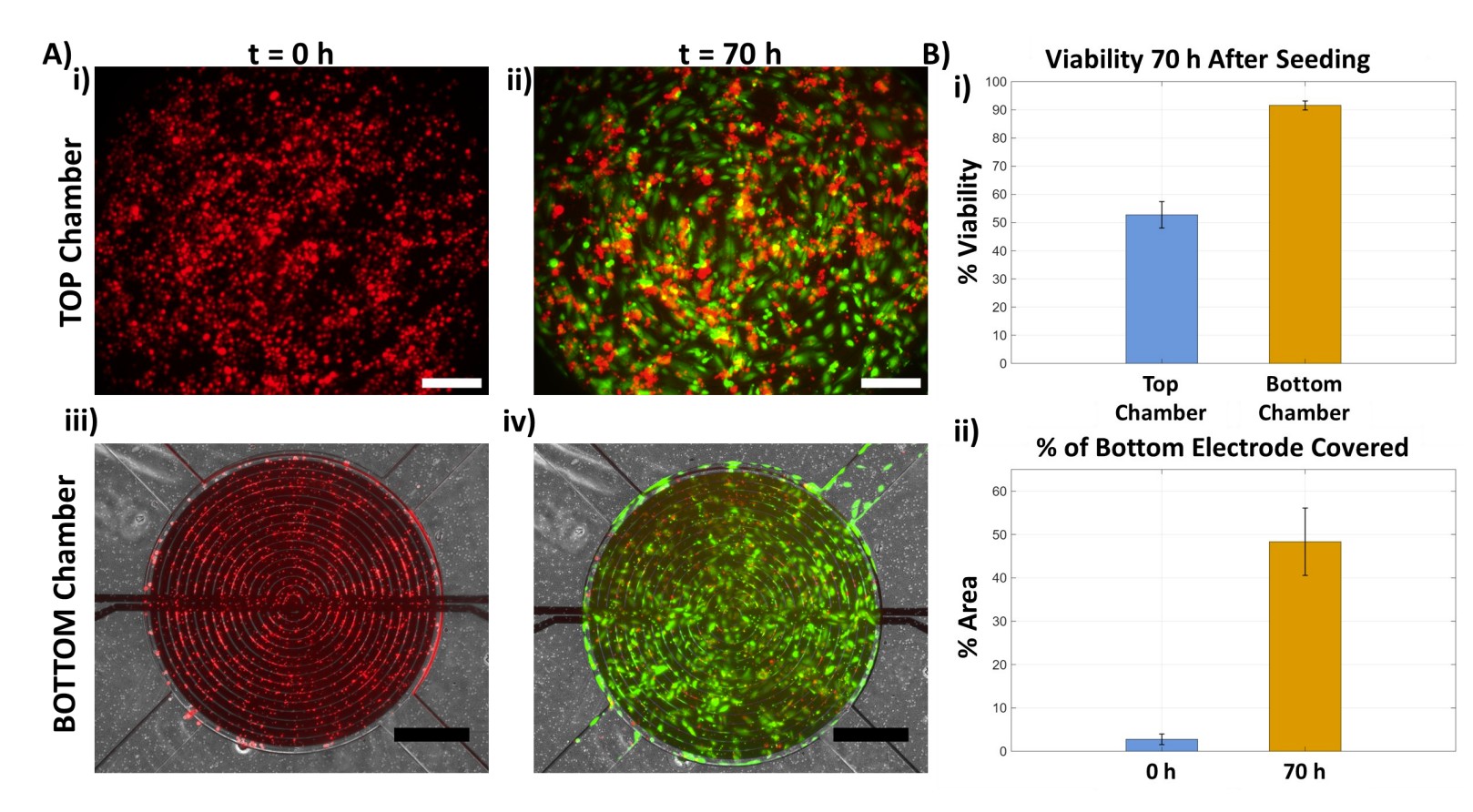
Figure 4. CoCM index experimental results at 30 kHz. Top chamber impedance (blue) decreases ~20% while bottom chamber (orange) increases ~15%. Symmetrical trends appear after 40 h, confirmed by fluorescence imaging (CellTracker Red). From Torres-Castro et al., Lab Chip, 2026.
Fluorescence validation
Parallel time-lapse fluorescence microscopy confirmed the impedance data. At t=0, almost no cells appeared in focus on the bottom electrode surface. After 45 hours, cells were clearly visible, overlaying the interdigitated electrodes. A calcein AM viability assay at 70 hours confirmed >90% viability in the bottom chamber, demonstrating that the impedance measurements (30 kHz AC signal) did not compromise cell health.
Enhanced sensitivity from small detection volume
The microfluidic format provides a surface area-to-detection volume ratio (SA/V) of approximately 0.005 µm⁻¹, which enhances interaction between the impedance sensors and migrating cells. This enabled detection of impedance changes from small numbers of cells, particularly in the bottom chamber where cells formed a monolayer with tighter standard deviations.
FLEXDYM vs. PDMS: Why the Material Choice Matters for Long-Term Cell Culture
The NIST researchers specifically selected FLEXDYM over PDMS for the CoCM device. In the paper, they highlight that FLEXDYM’s low gas permeability was critical to slowing medium evaporation during the 70-hour experiment, a common challenge in microfluidic long-term culture. Here’s how the two materials compare:
| Property | FLEXDYM™ | Traditional PDMS |
|---|---|---|
| Gas permeability | ✓ Low : reduces medium evaporation in multi-day experiments | ⚠ High : significant evaporation, frequent media changes needed |
| Bonding | ✓ Thermal bonding at 80 °C, no plasma treatment required | ⚠ O₂ plasma bonding (irreversible, time-sensitive window) |
| Pressure resistance | ✓ Withstands pressurized pump-driven flows | ⚠ Can delaminate under sustained pressure |
| Molecular sorption | ✓ Low absorption of small molecules from media | ⚠ Known absorption of hydrophobic compounds |
| Biocompatibility | ✓ ISO 10993 & USP Class VI certified | Generally biocompatible, not standardized for medical use |
| Scalability | ✓ Hot embossing, injection molding, roll-to-roll | ⚠ Casting-based, difficult to scale beyond lab prototyping |
| Validated cell types | Neurons, hepatocytes, endothelial, dermal, stem cells, iPSCs, 786-O carcinoma (see list) | Broadly validated but with caveats on sorption |
For detailed fabrication protocols with FLEXDYM and SUBLYM, see: FLEXDYM for Fast & Easy Microfluidic Device Fabrication and SUBLYM: 2-in-1 Machine for Hot Embossing and Bonding.
Applications: From Cancer Research to Organ-on-Chip
The CoCM platform’s combination of real-time dual-sided impedance, label-free detection, and microfluidic flow compatibility opens applications across multiple research domains:
Cancer metastasis research. Quantitative monitoring of tumor cell invasion through tissue barrier models. The use of 786-O renal carcinoma cells in this study directly demonstrates the platform’s relevance for studying invasive cancer behavior.
Organ-on-chip integration. The authors note that the platform is currently being adapted for heart-on-chip applications at NIST, with pump-driven flow for physiological shear stress. The FLEXDYM channels are designed to withstand the pressurized conditions required for these models. Similar integration is possible for lung-on-chip (with air-liquid interface) and other MPS architectures.
Drug screening. Real-time impedance readout enables quantitative assessment of how drugs affect cell motility, barrier permeability, and transendothelial migration, with temporal resolution not achievable by endpoint assays.
Immune cell dynamics. Studying leukocyte transmigration, inflammatory responses, and immune cell-tumor interactions at tissue interfaces, all processes that require real-time, label-free detection.
Scale-up for diagnostics. The thermoplastic fabrication approach (SUBLYM + FLEXDYM) is inherently scalable via injection molding and roll-to-roll embossing, making this architecture translatable to diagnostic cartridge manufacturing.
Eden Microfluidics: Products Used & Complementary Solutions
Two Eden products were used in this study. The rest of the product suite can complement and simplify the workflow for labs looking to replicate or extend this approach.
Frequently Asked Questions
Cross-over Cell Migration (CoCM) is a dual-sided impedance measurement method developed by NIST researchers (Torres-Castro et al., Lab on a Chip, 2026). It uses interdigitated gold electrodes patterned on both sides of a porous PET membrane to simultaneously monitor cell migration in real-time, tracking cells as they leave one surface and arrive on the other. The CoCM index is a normalization metric that enables direct comparison of impedance changes between chambers, accounting for baseline variability between instruments.
Cells adhering to or crossing interdigitated electrodes alter local impedance by changing the charge transfer resistance and capacitance at the electrode surface. In the CoCM platform, measurements at 30 kHz every 20 minutes detect these changes. As cells depart the top surface, impedance falls; as they arrive on the bottom, it rises. These inversely proportional trends quantify migration dynamics in real-time without labels or chemical modification.
FLEXDYM is a biocompatible thermoplastic copolymer (ISO 10993, USP Class VI) developed by Eden Tech. Compared to PDMS, it offers lower gas permeability (critical for multi-day cell culture), thermal bonding at 80 °C without plasma, pressure resistance for pump-driven flows, low molecular sorption, and compatibility with manufacturing scale-up via hot embossing and injection molding.
CoCM monitors both membrane sides simultaneously (vs. one-sided detection in transwells), requires no gel-based seeding, operates in a microfluidic format compatible with pump-driven organ-on-chip systems, and provides higher sensitivity thanks to the small detection volume (SA/V ~0.005 µm⁻¹). It also allows parallel optical imaging for multimodal validation.
The initial study used 786-O human renal carcinoma cells (adenocarcinoma, from ATCC), seeded at 2×10⁶ cells/mL, achieving >90% viability after 70 hours. The authors note the platform is applicable to cancer cells, immune cells, and other migratory cell types, and is being adapted for heart-on-chip applications at NIST. FLEXDYM itself has been validated with neurons, hepatocytes, endothelial cells, stem cells, and iPSCs (see published studies).
The SUBLYM is a compact, portable hot embossing machine from Eden Microfluidics. It uses controlled heat and pressure to transfer channel patterns from a master mold into thermoplastic sheets. In the NIST study, it operated at 170 °C for 360 s for embossing and 80 °C for 1 min for thermal bonding — functioning as a 2-in-1 embossing and bonding tool. It works with FLEXDYM, COC, PMMA, and PS.
Yes. The thermoplastic approach is inherently scalable. FLEXDYM is compatible with hot embossing for prototyping, roll-to-roll embossing for medium volume, and injection molding for mass production. The SUBLYM handles both embossing and bonding, reducing equipment needs. Eden also offers the Point-of-Care Scale-Up Kit for complete prototyping-to-production workflows.
Want to build a similar microfluidic platform?
Whether you're studying cell migration, building organ-on-chip models, or developing diagnostic devices, our team can help you go from design to working device.
Start Designing with FLUI'DEVICE Explore Materials & Equipment
